|
12/29/2023 0 Comments Endurance catheter Throughout catheter insertion, blood is contained within the device to aid in prevention of blood exposure. Remove 3 saline and 3 heparin syringes from their packages. With the option to treat patients with occluded midline catheters rather than replace, clinicians can reduce cost, increase patient satisfaction, and preserve vein health. The ARROW Endurance catheter system is a sterile, single use peripheral intravascular device designed to permit access to the peripheral vascular system. The catheter should be flushed every 24 hours and after each use. With this research, safety and efficacy appears to be established for 1 mg of alteplase, repeat times 1 mg if necessary, in midline catheters. Treating occluded central vascular access devices with 2 mg of alteplase has been safe and effective since 2001. None of the 112 patients had a major adverse event, and only 1 minor event was recorded and was resolved without removal of the midline catheter. Of those studied, 112 devices were treated with 1 mg of alteplase, and 109 had patency restored with 1 dose. Anchor the catheter to the skin using Steri-strips, sterile tape, or securement device Apply transparent permeable adhesive dressing (Tegaderm). A prospective open label study of 497 midline catheters was conducted from July 2018 to December 2018. The purpose of this research was to show evidence that alteplase, a US Food and Drug Administration-approved thrombolytic for central vascular access devices, is a safe and effective thrombolytic for midline catheters. 
Single-use peripheral intravascular device designed to permit access to the peripheral vascular system. Avoid freezing and excessive heat above 40☌ (104☏). A systematic review of ultrasound-accelerated catheter-directed thrombolysis in the treatment of deep vein thrombosis J Thromb Thrombolysis. 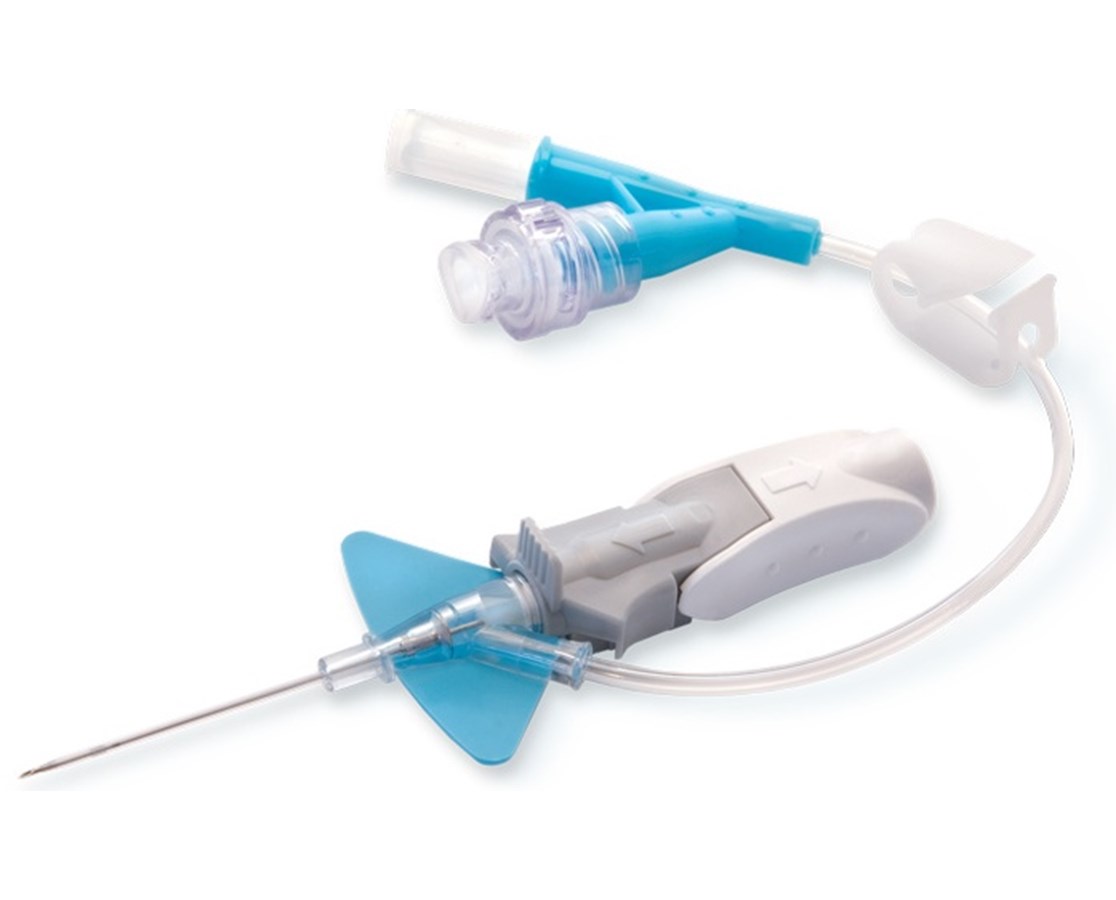
2 (2020-12) ARROW ENDURANCE Extended Dwell Peripheral Catheter System Store between 15 - 30☌ (59 - 86☏). Fluid infusion was immediately stopped, and the catheter was removed when the surgery was completed. ARROW ENDURANCE Extended Dwell Peripheral Catheter System EDC-00820-201A, Rev. This item may require 7-10 Business Days to process and prepare for shipping.Product Information: CATHETER, EXT DWELL ENDURANCE BASIC 20GX6CM (5/CS) Basic. 
Our packaging expertise allows us to offer comprehensive standard maximal barrier kits in the Arrow. We offer catheter options for both short-term and long-term dwell, antimicrobial and antithrombogenic protection, and pressure rated catheters. Currently, clinicians may not use this device as often as clinically indicated because they lack an approved intervention to treat thrombotic occlusions. Arrow Endurance Extended Dwell Peripheral Catheter System. Furthermore, the catheter was apparent in the aortic arch on TEE (Fig. In Central Access, we offer a broad range of catheter lengths, flow rates and customizable kits. Occluded catheters may be responsible for interruptions in therapy, delays in discharge, or additional procedures, such as catheter replacement. Price Action: TFX shares are up 0.29% at $237.98 on the last check Monday.Maintaining and restoring patency in midline catheters has been a significant issue in the hospitalized patient requiring a multitude of infusates and frequent blood specimen collection. Ensuring catheter patency is vital to delivering life-sustaining therapies Central line function should be routinely assessed. The midline catheters can remain in-situ up to 30 days. There have been 18 reported injuries and no deaths. According to Infusion Nurses Society, midline catheter is defined as a vascular access device which is inserted into a peripheral vein (namely, basilic, cephalic, or brachial vein), and the tip of it is at or near axilla level, and distal to the shoulder. 
Teleflex/Arrow International reports 83 complaints related to this issue. The issue may cause serious injury, including blockage of blood vessels, inadequate blood flow, injury to blood vessel walls, blood clots, blockage of the lung arteries (pulmonary embolism), heart attack, or death. The FDA writes that if the catheter separates while in a blood vessel, the catheter fragments could be left in the bloodstream and may migrate to other places in the body.Īlso Read: Recall Alert: Teleflex Recalls Rüsch Endotracheal Tubes Due to Connector Issues. The ARROW Endurance Extended Dwell Peripheral Catheter System allows healthcare providers access to a patient’s peripheral vascular system for short-term use to sample blood, monitor blood pressure, or administer fluids, blood, and blood products. The FDA has identified this as a Class I recall, the most serious type of recall. Teleflex Incorporated (NYSE: TFX) and its subsidiary Arrow International are recalling the ARROW Endurance Extended Dwell Peripheral Catheter System after reports of catheter separation or leakage. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
